24 Apr, 2026
Feel free to reach out to us.

24 Apr, 2026
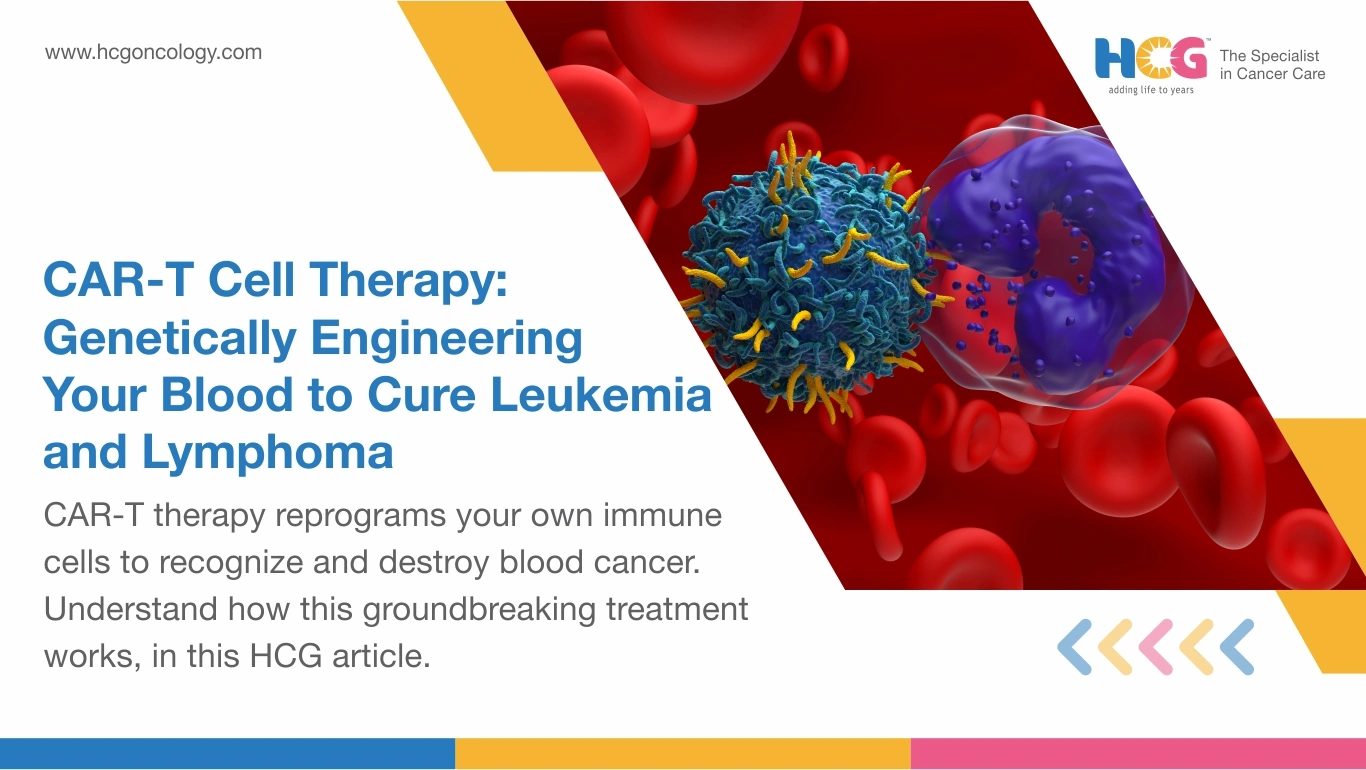
When every treatment has been tried, and the cancer still comes back, families find themselves asking a question nobody wants to face: Is there anything left? For patients with relapsed leukemia or lymphoma, CAR T-cell therapy has become a genuine answer to that question. A uniqe treatment for certain blood cancers, CAR T-cell therapy takes your own T-cells, rewires them in a laboratory to seek and destroy cancer cells, then releases them back into your bloodstream as a living, self-multiplying treatment. One infusion. Cells that keep working for months, sometimes years. In India today, this is not a distant option. It is happening.
Most people picture the immune system as a guard that fights anything foreign. Cancer survives specifically because it stops looking foreign. It learns the body's language, wears the right disguise, and quietly multiplies while every natural alarm stays silent.
CAR T-cell therapy tears at the genetic level. Your harvested T-cells are fitted with a chimeric antigen receptor, an engineered molecular lock built to recognize one specific protein on cancer cell surfaces. In B-cell blood cancers, that protein is CD19. When a reprogrammed T-cell meets a CD19-carrying cancer cell, the recognition is instant. The destruction follows immediately. Every healthy cell nearby is left completely untouched.
What makes this therapy genuinely different is what happens after infusion. These modified cells do not wear out and disappear. They replicate. From a single treatment session, a patient's body builds a sustained, self-renewing population of cancer-targeting cells.
It is important to note that unlike chemotherapy or targeted drugs taken repeatedly, one CAR T-cell therapy infusion can generate immune activity that persists for years.
| Step | What Happens | Timeframe |
|---|---|---|
| 1. Harvest | T-cells withdrawn via a leukapheresis machine | 3–5 hours |
| 2. Engineer | Viral vector transduction genetically modifies the cells | 2–4 weeks |
| 3. Lymphodepletion | A short chemotherapy course clears the immune space | 3–5 days |
| 4. Infuse and Monitor | Engineered cells returned; inpatient observation begins | 7–14 days |
At HCG's Hemato-Oncology Unit, every stage runs under one connected clinical team. Apheresis scheduling, laboratory coordination, and ICU monitoring are managed together, never handed between separate departments.
Not all patients with blood cancer can receive CAR T-cell therapy. CAR T-cell therapy is considered for patients whose cancer has returned or stopped responding after at least two prior treatment courses. Qualifying diagnoses include relapsed diffuse large B-cell lymphoma, relapsed or refractory ALL in adults and children, and certain myeloma cases.
Kidney function, liver health, heart stability, absence of active infection, and physical resilience all form part of the assessment. At HCG, every referral is reviewed individually. There is no standard template for eligibility.
Note: CAR T-cell therapy and bone marrow transplants are entirely different procedures. A transplant rebuilds the blood-forming system using someone else's cells, carrying the risk of graft-versus-host disease. CAR T-cell therapy uses only your own cells. That risk simply does not exist here.
Cytokine Release Syndrome (CRS)is the side effect that clinical teams track from the first moment after infusion. When engineered T-cells start destroying cancer cells at speed, the immune system responds with a full inflammatory surge. Mild CRS feels like being hit hard by the flu: fever that will not break, exhaustion that reaches into the bones, and aching muscles throughout the body. Severe CRS is a different situation entirely, bringing blood pressure instability, respiratory distress, and organ stress happening simultaneously.
ICANS (Immune Effector Cell-Associated Neurotoxicity Syndrome) shows up as neurological disruption. Patients describe it as thinking through fog, words that refuse to come, and, in rare cases, seizure activity.
Usually, specialists deploy tocilizumab, a targeted IL-6 receptor blocker, to interrupt the inflammatory cascade before it reaches dangerous severity. Monitoring is not scheduled at intervals. It runs continuously from the infusion onward.
In summary, CAR T-cell therapy is not safe at centers without dedicated cellular therapy infrastructure. The side effect profile is too serious for improvised management.
Around 30-40% of relapsed large B-cell lymphoma patients achieve complete remissions lasting beyond two years. Before CAR T-cell therapy existed, those same patients had a median survival of months on salvage chemotherapy.
In relapsed ALL, complete remission rates cross 70-80% across documented clinical cohorts. Overall lymphoma response rates run between 52% and 82%, varying by product and individual patient profile. Every person in those statistics had run out of conventional options beforehand.
CAR T-cell therapy cost in India can go up to Rs. 35,00,000. Some hospitals have CAR T-cell therapy packages, which patients can opt for.
Costs vary by hospital and patient profile. The infrastructure this therapy requires exists primarily in Bangalore, Kolkata, and Mumbai. Government scheme inclusion is slowly expanding. HCG's financial counseling team works with each family individually to find every available support route.
The first two weeks after infusion are the hardest medically. CRS and ICANS surveillance runs continuously throughout. After discharge, the work is not over. CD19-targeting clears healthy B-cells alongside cancerous ones, which means immunoglobulin replacement and careful infection management continue for months.
The fatigue during this recovery period is not ordinary tiredness. It settles deeply into the body and resists quick fixes. Structured nutrition, psychological support, and realistic expectations matter as much as clinical monitoring. PET-CT imaging at 30 and 90 days maps remission depth. HCG's onco-psychology team remains available throughout because recovering from CAR T-cell therapy is not a purely physical process.
Full immune reconstitution takes 6-12 months. Build the recovery plan around that reality from the very beginning, not after discharge.
When decisions need to be made, HCG helps by treating CAR T-cell therapy as a continuous clinical relationship, not a single procedure with a start and end date. The hemato-oncology teams across various hospitals of the HCG network accompany patients from the first eligibility conversation through every stage: apheresis, infusion, post-infusion monitoring, and long-term survivorship follow-up.
When you visit the doctor to discuss CAR T-cell therapy as a treatment option:
Disclaimer: This information is intended to educate patients and caregivers. It does not replace professional medical advice. All treatment decisions should be made in consultation with a qualified doctor.