27 Apr, 2026
Feel free to reach out to us.

27 Apr, 2026
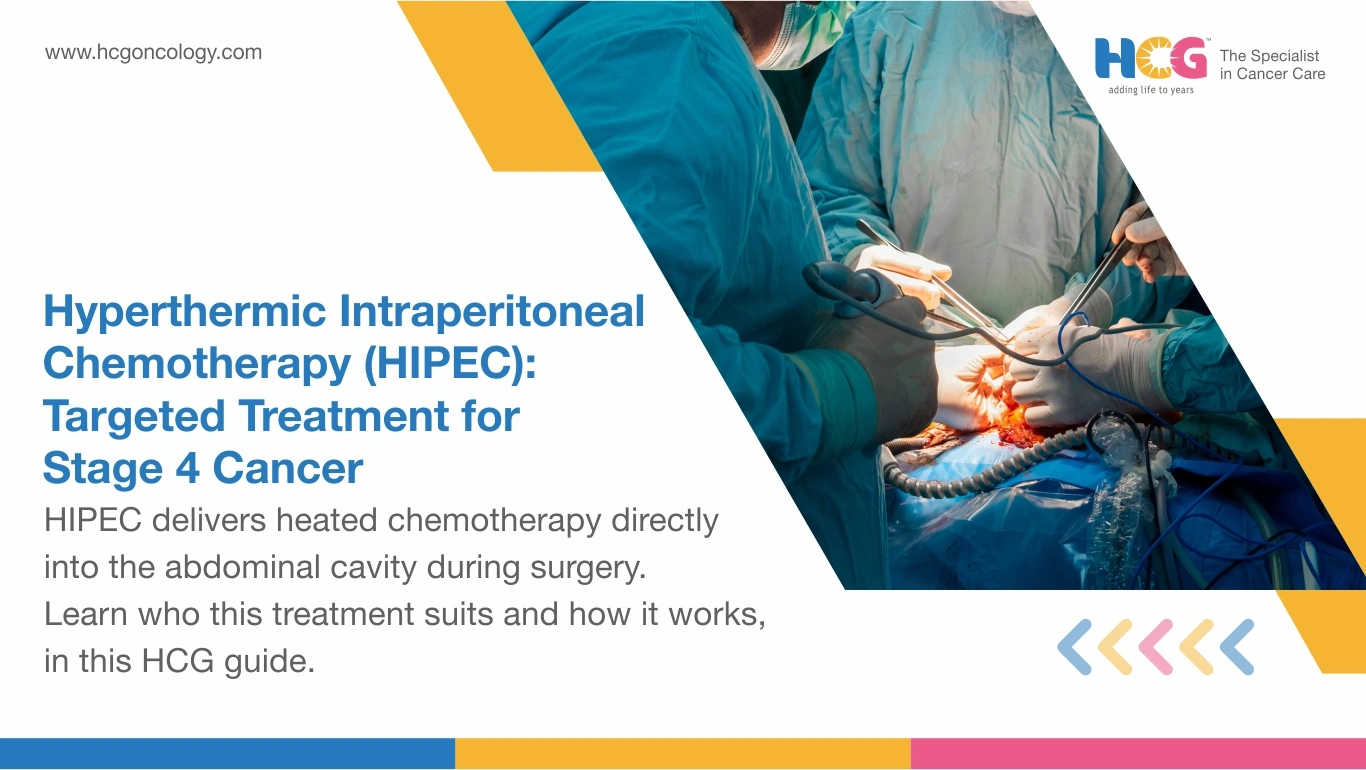
Hyperthermic Intraperitoneal Chemotherapy (HIPEC) treatment for cancer delivers highly concentrated, heated chemotherapy directly into the abdominal cavity immediately after surgical tumor removal. By raising drug temperature to 41-43°C, chemotherapy agents penetrate residual cancer cells invisible to the naked eye, improving local treatment effectiveness. Used for advanced cancers with peritoneal surface spread, such as ovarian, colorectal, appendix, and gastric primaries, HIPEC combined with cytoreductive surgery (CRS) significantly extends survival beyond standard systemic chemotherapy alone.
HIPEC treatment for cancer is a two-stage intraoperative procedure combining maximal surgical debulking with direct peritoneal chemotherapy delivery. Cytoreductive surgery systematically removes all visible peritoneal tumor implants. Right after, a heated chemotherapy solution is circulated through the peritoneal cavity for 60 to 90 minutes while the patient remains on the operating table.
Intravenous chemotherapy reaches the peritoneum at diluted systemic concentrations after passing through the entire circulatory system. HIPEC bypasses this dilution entirely, flooding the peritoneal surface with significantly higher local drug concentrations, enhancing the overall effectiveness of the treatment.
| Feature | Standard IV Chemotherapy | HIPEC |
|---|---|---|
| Delivery method | Intravenous infusion | Direct intraperitoneal circulation |
| Drug concentration | Low (systemic dilution) | Significantly higher local concentration |
| Target | Systemic disease | Peritoneal surface microscopic residue |
| Temperature | Room temperature | 41–43°C heated solution |
| Timing | Multiple cycles over months | Single intraoperative session |
HIPEC treatment for cancer is effective for stage 4 cancers, specifically confined to the peritoneal cavity without distant organ metastases. Stage 4 cancer with isolated peritoneal carcinomatosis and no liver, lung, or bone metastasis represents a fundamentally different surgical landscape from widespread systemic disease (NCBI).
A report from Hopkins Medicine says that for appendix cancer with peritoneal spread, CRS and HIPEC achieve a median survival of 30 to 60 months compared to 8 to 12 months with systemic chemotherapy alone (NCBI). For colorectal cancer peritoneal metastasis, selected patients achieve five-year survival rates of 30 to 45% following CRS and HIPEC, outcomes not achievable with systemic therapy alone.
In summary, HIPEC is not a universal stage 4 cancer treatment. Its survival benefit is specific to peritoneal surface malignancies in patients whose disease has not spread beyond the abdominal cavity.
Appendiceal cancer (pseudomyxoma peritonei) is the strongest HIPEC indication globally, with the highest published survival benefit. Appendix cancer spreads primarily by seeding the peritoneal surface rather than through the bloodstream, making peritoneal-directed therapy biologically ideal (Mayo Clinic).
Ovarian cancer with peritoneal spread is the second major indication. HIPEC following interval debulking surgery improves recurrence-free survival in eligible patients with platinum-sensitive disease.
Colorectal cancer with peritoneal metastasis is eligible when the Peritoneal Cancer Index (PCI) score is below 20. Gastric cancer peritoneal spread and mesothelioma represent additional indications with stricter patient selection criteria.
It is also recommended for selected gastric cancer and peritoneal mesothelioma cases.
CRS and HIPEC are among the most technically demanding oncological procedures available. The surgical pathway at HCG's HIPEC Surgery program follows six structured stages:
Total operative time ranges from 8 to 14 hours. HIPEC procedures at HCG Cancer Hospital are performed by a dedicated PSM surgical team with specialist perioperative nursing and anesthesiology support.
HIPEC side effects differ meaningfully from standard intravenous chemotherapy. Systemic toxicity is significantly lower because chemotherapy is regionally delivered rather than circulating through the full bloodstream. Severe hair loss, prolonged bone marrow suppression, and systemic mucositis are substantially reduced.
The primary HIPEC-specific risks relate to surgical complexity, namely anastomotic leak, intra-abdominal infection, pleural effusion, and prolonged ileus, which are the clinically significant postoperative complications requiring monitoring. Renal function monitoring is critical when cisplatin-based regimens are used
Recovery after CRS and HIPEC requires 10 to 21 days of inpatient monitoring before discharge, with most patients hospitalized for 14 days on average (Mayo Clinic).
Days 1 to 3:ICU monitoring for hemodynamic stability, renal function, and anastomotic integrity. Parenteral nutrition support during this phase.
Days 4 to 7:Transition to surgical ward. Gradual bowel function return assessed through flatus and bowel sounds.
Days 8 to 14:Dietary advancement from clear liquids to soft solids. Wound care, drain management, and mobilization physiotherapy were initiated.
Weeks 3 to 6:Return to light daily activities. Full functional recovery requires 6 to 12 weeks. Nutritional rehabilitation with a clinical dietitian continues throughout.
Post-discharge CBC monitoring, liver function tests, and wound healing assessment are run at two- to four-week intervals. CT surveillance for recurrence begins at three months post-HIPEC. Psycho-oncology support and fatigue management are integrated into HCG's post-HIPEC survivorship pathway.
The combined CRS and HIPEC procedure ranges from Rs. 6,00,000 to Rs. 20,00,000, depending on cytoreductive complexity, organs resected, and ICU stay requirements.
Costs vary by hospital and patient profile. Bangalore, Mumbai, Delhi, and Chennai carry higher facility charges than Tier 2 and Tier 3 city hospitals. Ayushman Bharat and CGHS coverage apply to eligible patients. HCG's financial counseling team maps financial support pathways before surgical planning begins.
HCG Cancer Hospital approaches this by ensuring patients with peritoneal surface malignancies receive a rigorous, evidence-based eligibility assessment before CRS and HIPEC are recommended. HCG's HIPEC Surgery program combines high-volume cytoreductive surgical expertise with dedicated perioperative care and post-HIPEC survivorship support. For patients with appendix cancer, ovarian cancer, peritoneal spread, or colorectal peritoneal metastasis meeting PCI and performance criteria, CRS and HIPEC represent the most effective treatment strategy for meaningful long-term survival.
Next Steps for Your Doctor Visit:
Disclaimer: This information is intended to educate patients and caregivers. It does not replace professional medical advice. All treatment decisions should be made in consultation with a qualified doctor.