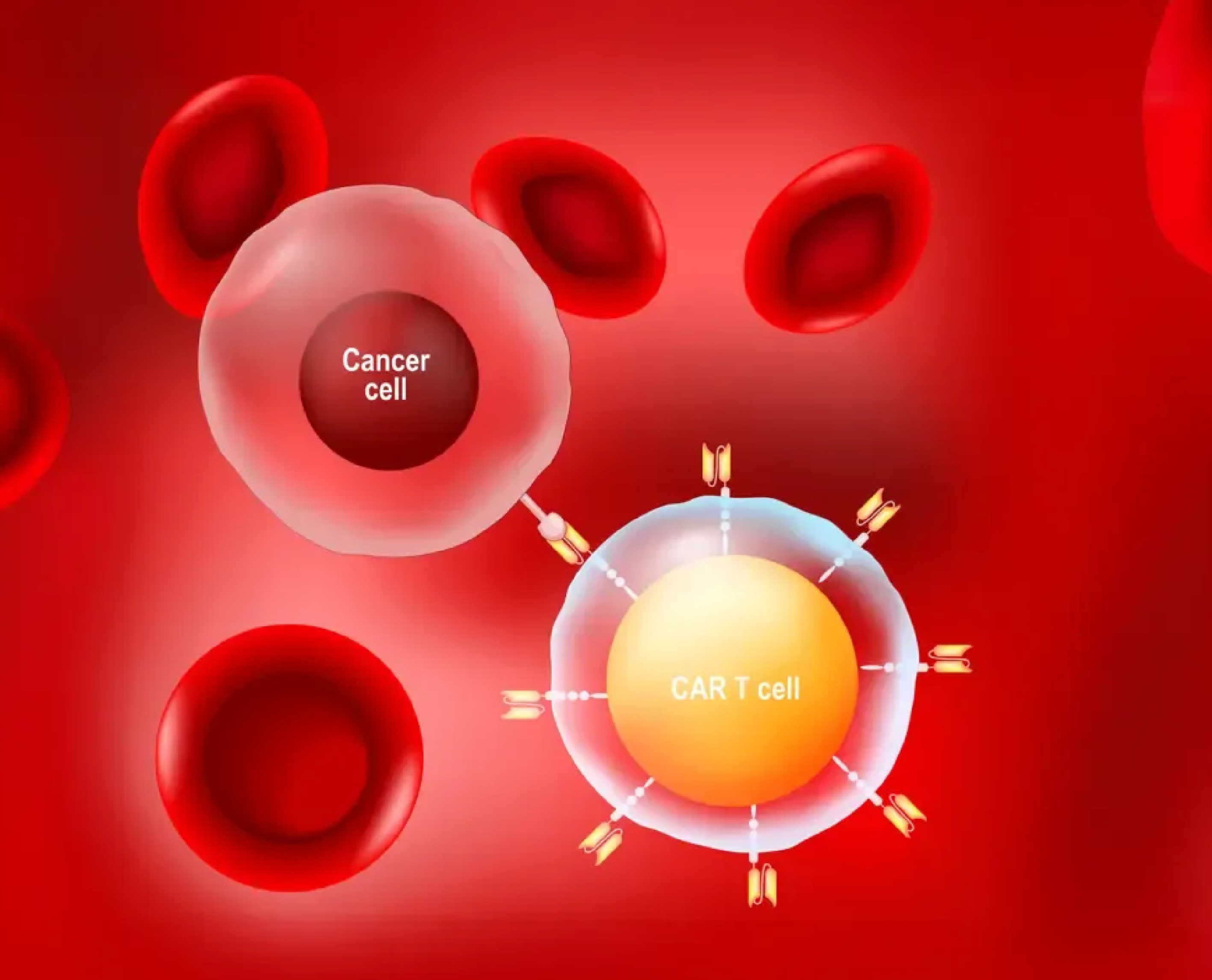
Introduction: Reprogramming the Immune System
CAR T-cell therapy involves collecting a patient's own T-cells and genetically modifying them in a laboratory setting to express Chimeric Antigen Receptors (CARs). These receptors are specifically designed to identify specific proteins found on cancer cells.
Once infused back into the body of the patient, the altered T-cells identify, bind to, and destroy cancer cells. To effectively target cancer cells, CAR T-cells are designed to persist for months or longer. CAR T-cell therapy is also referred to as a "living drug" because these modified T-cells multiply and are active inside the body for longer periods.
Studies show that CAR T-cell therapy is effective in treating some blood cancers that have relapsed or are resistant to treatment, particularly B-cell malignancies and multiple myeloma.
Antigen-specific CAR T-cell therapy treats cancer in a targeted manner, unlike chemotherapy, which targets both healthy and cancer cells.
The HCG Advantage
HCG has established itself as one of the best hospitals for CAR T-cell therapy in Bangalore by combining clinical experience in India with international hemato-oncology treatment standards. Our specialists place a special emphasis on guaranteeing safe delivery, implementing a systematic monitoring system, and responsibly choosing the right patients who can benefit from this treatment, such as those with relapsed or refractory B-cell malignancies, which are the primary indications for CAR T-cell therapy.




