05 May, 2026

05 May, 2026
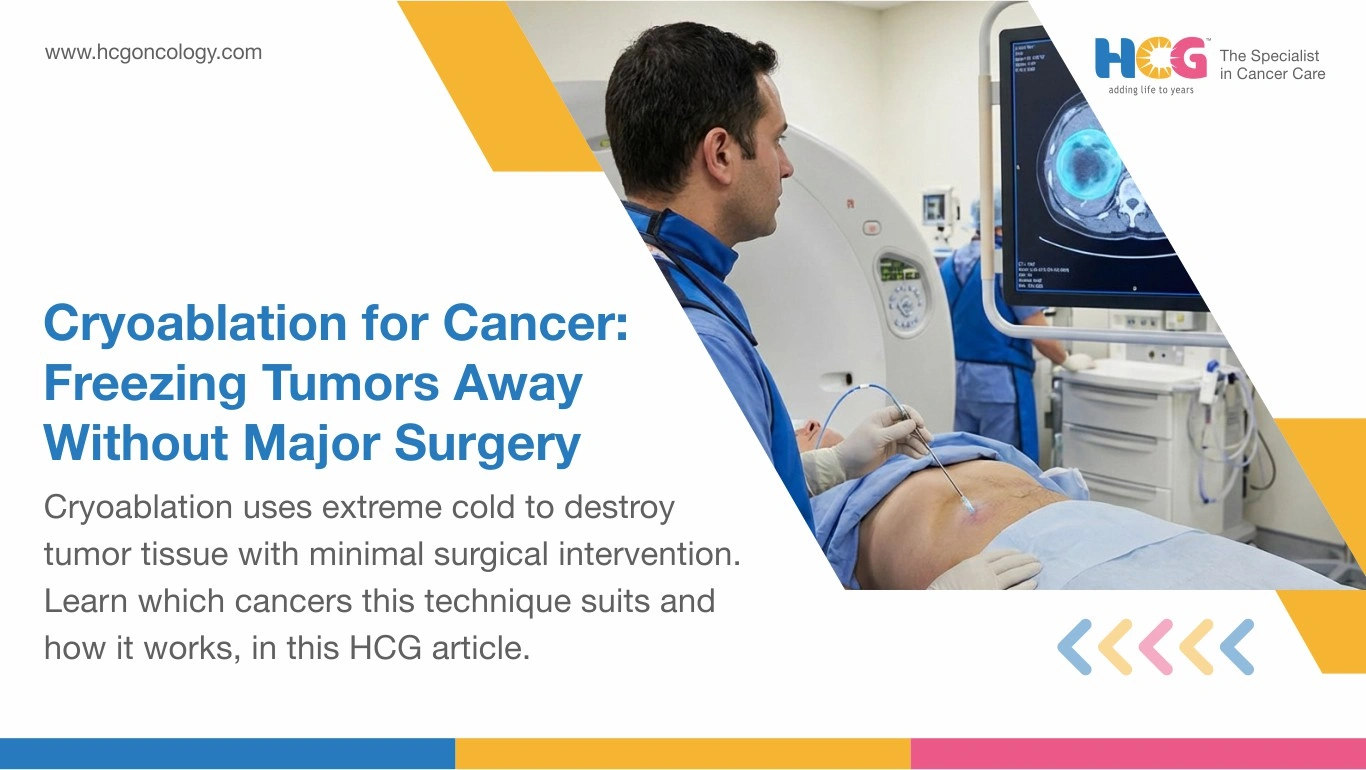
Cryoablation for cancer works by channeling extreme cold through a needle-thin probe directly into a tumor, crystallizing malignant cells from the inside out. No large incision. No prolonged hospitalization. A device called a cryoprobe delivers argon gas to the tumor site, generating temperatures far below freezing and forming a visible ice ball that obliterates the target tissue. Published data in Theranostics Journal confirm that cryoablation produces a superior tumor response and faster recovery compared to several heat-based ablation alternatives. For carefully selected patients, it is a clinically sound path to nonsurgical tumor removal.
Cell death during cryoablation for cancer happens across two deliberate phases.
First, argon gas chills the cryoprobe tip below -100 degrees Celsius, growing an ice ball that ruptures cell walls through intracellular crystal formation.
Second, a helium-gas thaw cycle reverses the temperature. The osmotic shock from subsequent refreezing triggers secondary cellular collapse.
Most sessions involve two complete freeze-thaw cycles to maximize ablation completeness.
The table below compares open surgery and cryoablation across key clinical factors:
| Factor | Open Surgery | Cryoablation |
|---|---|---|
| Incision Size | Several centimeters | 1.5 mm probe entry |
| Anesthesia | General | Local or conscious sedation |
| Hospital Stay | 3 to 7 days | Often same-day discharge |
| Recovery Time | 4 to 6 weeks | 1 to 2 weeks typically |
| Bleeding Risk | Higher | Substantially lower |
Cryoblation is recommended for different types of cancer, depending on the severity of the disease and individual case parameters:
| Cancer Type | How Cryoablation Helps |
|---|---|
| Prostate Cancer | Freezes and destroys localized tumor tissue; used as primary treatment or after failed radiation therapy. |
| Kidney Cancer | Targets small renal masses under 4 cm; preserves surrounding kidney function better than surgical removal. |
| Breast Cancer | Eliminates small, early-stage tumors under 2 cm in an outpatient setting without the need for open surgery. |
| Lung Cancer | Treats inoperable NSCLC tumors; also relieves airway obstruction and may enhance response to immunotherapy. |
| Liver Cancer | Controls hepatocellular carcinoma and liver metastases that cannot be surgically removed; can be curative for limited disease. |
| Bone Tumors | Destroys tumor tissue and provides significant pain relief in both benign bone tumors and metastatic bone lesions. |
| Cervical Precancer | Freezes abnormal cervical cells (CIN) in an outpatient procedure, preventing progression to invasive cancer. |
| Colorectal Cancer (Metastatic) | Treats liver and lung metastases; used alongside chemotherapy to improve outcomes in limited metastatic disease. |
| Soft Tissue Sarcomas | Manages surgically inaccessible tumors; controls disease burden and relieves symptoms in advanced or recurrent cases. |
| Skin Cancer | Liquid nitrogen applied directly destroys basal cell carcinoma, squamous cell carcinoma, and precancerous skin lesions. |
Tumor size, precise anatomical location relative to blood vessels and nerves, and the patient's baseline functional status all determine whether cryoablation is appropriate. A multidisciplinary review is standard before scheduling.
Discomfort during the procedure is minimal. Local anesthesia or conscious sedation is used to manage probe insertion. Post-procedure, patients typically notice a dull, pressure-like ache at the treated site for 24 to 48 hours, which oral analgesics can effectively relieve.
The freeze zone carries a mild anesthetic quality, so intraoperative pain is generally low. Patients treated for bone metastases often report meaningful pain reduction within days, which is a well-documented secondary benefit of cryoablation beyond tumor destruction.
Heat-based methods like radiofrequency ablation and microwave ablation carry the risk of thermal injury to bile ducts, vessels, and nerves adjacent to the tumor. Cryoablation avoids this by creating a precisely visible, cold ablation zone under continuous imaging surveillance.
The reality is, interventional radiologists prefer cryoablation near thermally sensitive structures precisely because the ice ball boundary is unambiguous on CT or ultrasound. Comparative data from a 2022 study published in the Theranostics Journal confirms quicker recovery times alongside strong ablation efficacy.
Evolving research also documents that cryo-treated tumors release tumor antigens into the bloodstream after cell death, potentially triggering systemic anti-tumor immune activity. This combination approach, termed “cryo-immunotherapy,” is under active clinical investigation.
After completing cryoblation treatment, cancer patients need to adhere to the recovery and follow-up guidelines shared by doctors. Here is what the post-procedure period involves in practice:
When decisions need to be made, HCG helps by bringing together interventional radiologists, surgical oncologists, and medical oncologists to determine whether cryoablation for cancer fits a patient's specific tumor profile and treatment goals. Doctors carefully assess the eligibility of patients before recommending this procedure. Every case is mapped through imaging, reviewed by the multidisciplinary team, and planned with recovery in mind. If you are considering non-surgical tumor removal, HCG Cancer Hospital's interventional oncology team is fully equipped to help you manage your condition and support your well-being.
Disclaimer:This information is intended to educate patients and caregivers. It does not replace professional medical advice. All treatment decisions should be made in consultation with a qualified doctor.
Feel free to reach out to us.